Aruncus L. is a small genus distributed in the circumpolar regions of the Northern Hemisphere, including Europe, Asia, and North America. Number of species in this genus varies depending on the author, with some recognizing one highly polymorphic species of A. dioicus (Walter) Fernald (Hara, 1955) or the three species of A. dioicus, A. gombalanus (Hand.-Mazz.) Hand.-Mazz., and A. aethusifolius (H. Lév.) Nakai (Gu and Alexander, 2003; Lee, 2007). Aruncus is easily distinguished from other genera in Rosaceae by several characters, such as dioecious, perennial herbs, 2–3-pinnately or 1–2-ternately compound leaves without stipules, paniculate inflorescences, and follicular fruits (Hutchinson, 1964; Henrickson, 1985; Kalkman, 2004). Recent phylogenetic analyses suggested that Aruncus is sister to Holodiscus (K. Koch) Maxim., which is a small erect, hermaphroditic shrub in western North America within the tribe Spiraeeae (Potter et al., 2007a, 2007b; Xiang et al., 2016). Plants of Aruncus are widely cultivated as ornamentals (Hutchinson, 1964) and young leaves, with high antioxidant activities (Shin et al., 2008), are often consumed as vegetables.
In Korea, there are two taxa. Aruncus dioicus var. kamtschaticus is distributed in the higher elevations of mountains on the Korean Peninsula and on Ulleung Island, but it has not been reported on Jeju Island. This taxon is widely distributed in East Asia from western China, Korea, to Japan with a wide range of morphological variation (Ikeda, 2001; Gu and Alexander, 2003; Lee, 2003; Lee, 2007). Aruncus aethusifolius is an endemic species of Korea, occurring only on Jeju Island. It is usually found in moist places near or crevices of volcanic rocks at elevations ranging from 1,500 to 1,800 m as part of the subalpine vegetation on Mt. Halla, and it occurs as well on moist banks along creeks of the mountain at elevations as low as 400 m. Aruncus aethusifolius is occasionally thought of as a variety of A. dioicus (Hara, 1955; Lee, 2003), but it is considered to be a distinct species due to several unique morphological characteristics, such as a short plant stature, very short internodes in the stems, highly dissected leaflets, and erect follicles (Nakai, 1912; Lee, 1980; Lee, 2006; Lee, 2007).
One of key characters of Aruncus includes the sexuality of the species, in which staminate and carpellate flowers are developed in different individuals in a population, making it dioecious. Dioecious species is very rare among the approximately 90 genera and 3,000 species in Rosaceae, occurring only in eight genera and approximately nine species (Table 1). Given that the vast majority of species in Rosaceae are hermaphroditic and considering the phylogeny of Rosaceae (Potter et al., 2007a; Xiang et al., 2016), dioecy in Rosaceae were likely derived independently in each of the species from hermaphroditic ancestors. Dioecy is also rare in angiosperms, found in about 6% of flowering plants (Renner and Ricklefs, 1995), in contrast to the animal system, in which gonochorism with separate males and females is found in 95% of all species (Jarne and Auld, 2006).
The rarity of dioecism in angiosperms has led to much attention regarding the evolution and development of separate genders (Darwin, 1877; Westergaard, 1958; Irish and Nelson, 1989; Renner and Ricklefs, 1995; Sakai and Weller, 1999; Charlesworth, 2015; Käfer et al., 2017; Pannell, 2017; Zuluvova et al., 2017). Several characteristics, such as woodiness, wind pollination, and large inflorescences with small white to green flowers, are associated with the evolution of dioecy. With regard to their geographic distribution, dioecious species are more common in the tropics and on oceanic islands, including Hawaii, in angiosperms (Renner and Ricklefs, 1995; Sakai and Weller, 1999; Käfer et al., 2017). Aruncus provides an intriguing system with which to study the evolution of dioecy, as it deviates from the general trends in the evolution of the sexual system. However, there is confusion regarding the sexuality of Aruncus among authors. While most authors (Radford et al., 1964; Ohwi, 1965; Gleason and Cronquist, 1991; Ikeda, 2001; Lee, 2007; Mellichamp and Wetherwax, 2012; Mellichamp, 2014) have considered Aruncus to be dioecious, Gu and Alexander (2003) have found that Aruncus is monoecious, in which staminate and carpellate flowers are borne on the same plant. Tutin (1968) recognized that Aruncus is polygamo-dioecious, in which bisexual (or perfect) flowers are developed in some of the male or female plants in a population. Bond (1962) and Robertson (1974) showed that bisexual flowers are occasionally found in staminate inflorescences in A. dioicus var. dioicus in eastern North America, consistent with Tutin (1968). The occurrence of bisexual flowers is unknown in other varieties of A. dioicus and A. aethusifolius.
The objectives of this study are to characterize the sexuality of A. aethusifolius based on field observations and investigations of herbarium specimens and to provide insights into the evolution of sexual system of this species.
Materials and Methods
Field observations of A. aethusifolius were made on Mt. Hallasan on Jeju Island. Fifty-three herbarium specimens of A. aethusifolius and 190 specimens of A. dioicus var. kamtschaticus were examined in KB (the acronyms follow Thiers, 2017), TI, TUT, and Warm-Temperate and Subtropical Forest Research Center. A list of specimens of Aruncus aethusifolius examined in this study was provided in Appendix 1.
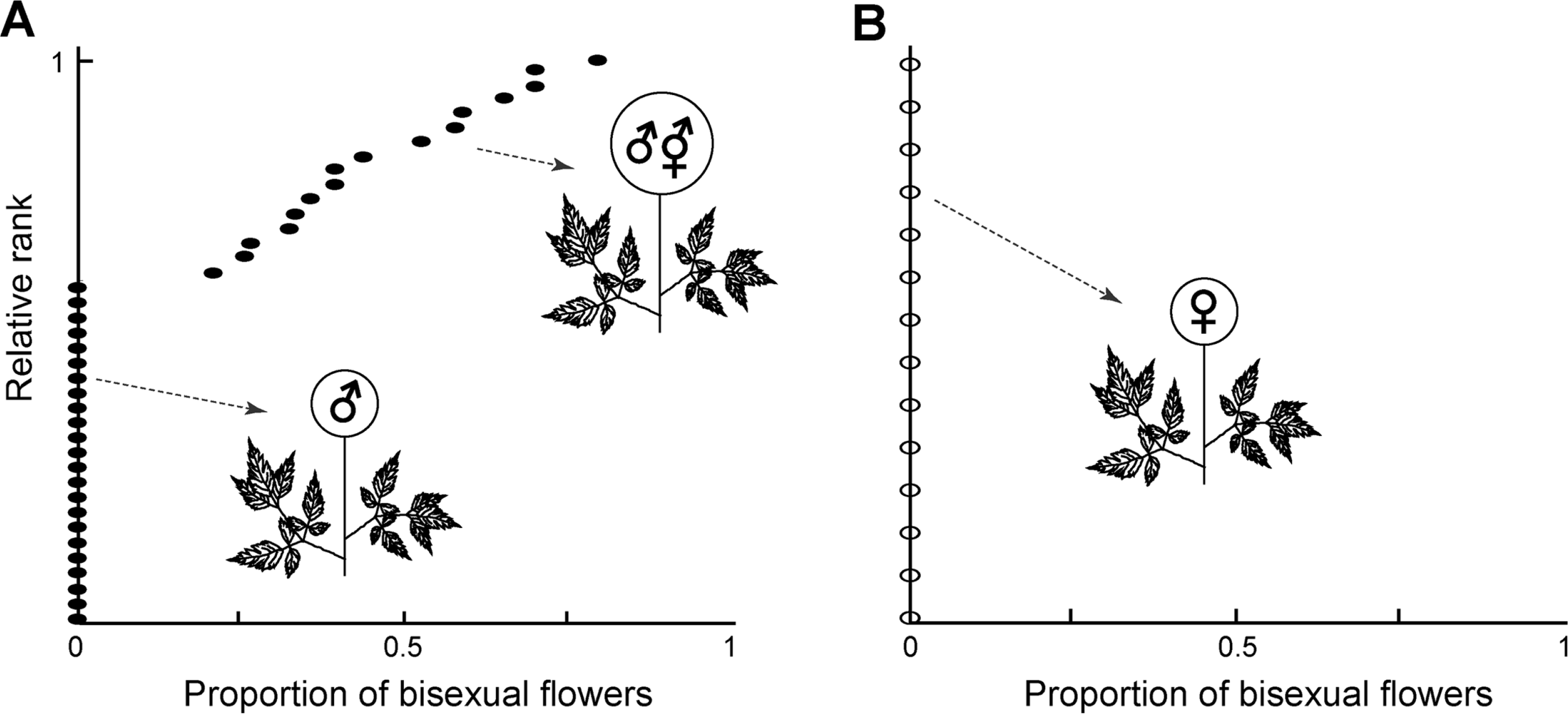
The sexuality of A. aethusifolius in a flower and a plant was determined under a microscope. A Nikon SMZ18 dissecting stereomicroscope (Nikon, Tokyo, Japan) was used to examine and photograph the flowers. Each specimen was scored as a male or female plant. The proportion of bisexual flowers in a plant was approximated by the length of the inflorescence axis, with bisexual flowers divided by the total length of the inflorescence axis. Flowers of A. aethusifolius are very small and borne very tightly along the inflorescence axis. The proportion of bisexual flowers in each gender class was plotted.
Results and Discussion
Our examination of A. aethusifolius indicated that the species has complex patterns of sexuality (Fig. 1). Three types of flowers were found: carpellate (Fig. 1B), staminate (Fig. 1D), and bisexual (Fig. 1F) flowers. The flowers had four basic whorls: sepals, petals, stamens, and carpels. In the carpellate flowers, stamens were not fully developed such that the filaments were very short and the anthers minute (Fig. 1B). In the staminate flowers, rudimentary carpels were located at the center of the flower (Fig. 1D). The bisexual flowers had both stamens and carpels that were fully developed (Fig. 1F). Nectaries on the inner side of the hypanthium were developed in all three types of flowers, which attract pollinators (Fig. 1). The flowers of A. aethusifolius are borne in panicles of racemes with 2–9 lateral branches. Carpellate flowers are always borne in separate individuals, making them female (gynoecious) plants (Fig. 1A). There are male (androecious) plants bearing only staminate flowers (Fig. 1C). In some male plants, bisexual flowers were also developed with staminate flowers in an inflorescence (Fig. 1E). Bisexual flowers are located at the lower part of a branch or at the lateral branch of the inflorescence. These andro-polygamous plants have mixed of flowers, staminate and bisexual flowers. The proportion of bisexual flowers in andro-polygamous plants based on herbarium specimens varied, ranging from 22 to 79% (Fig. 2). Thus, the populations of A. aethusifolius consist of female, male, and andro-polygamous plants. Of the 53 specimens examined, 14 were female, 23 were male, and 16 were andropolygamous (Fig. 2). As a result, sexuality of A. aethusifolius is defined as polygamo-dioecious.
Polygamo-dioecious may be a general condition in Aruncus. An examination of 190 specimens of A. dioicus var. kamtschaticus collected in Korea, China, and Japan showed that one specimen from Mt. Hwangbyeongsan in Gangwon Province (Kwon 080727-080, KB278766) was andropolygamous. The presence of andro-polygamous plants in A. dioicus var. kamtschaticus, albeit rare, is consistent with our observations of A. aethusifolius. Thus, our investigations of the sexuality in A. aethusifolius and A. dioicus var. kamtschaticus support previous studies of A. dioicus var. dioicus (Bond, 1962; Tutin, 1968; Robertson, 1974).
The occurrence of andro-polygamous plants in a dioecious species is occasionally reported in Rosaceae. For example, flowers of Oemleria cerasiformis (Torr. & A. Gray ex Hook. & Arn.) J. W. Landon are usually unisexual, developed in separate plants. However, in rare cases, bisexual flowers are also found (Hess, 2014), though the frequency of bisexual flowers is largely unknown.
The frequency of andro-polygamous plants varies across species in Aruncus. The occurrence of an andro-polygamous plant is very rare in A. dioicus var. kamtschaticus, as described above. It is difficult to quantify the frequency of andropolygamous in the eastern North American A. dioicus var. dioicus. Robertson (1974) stated that it was ‘occasionally’ found. However, the frequency of andro-polygamous plants in A. aethusifolius is much higher than the frequency in A. dioicus. Of the 53 specimens examined, 16 (30%) were andropolygamous (Fig. 2). The gender frequency based on herbarium specimens may be biased, as female and andro-polygamous plants could be over-represented. Female and andropolygamous plants bearing flowers or fruits could be prepared for specimens, while male plants may have been neglected when the flowers fell off. Our field study supports the underrepresentation of male plants in herbarium specimens. Among nearly 60 individuals of nine clumps of A. aethusifolius along the Youngsil trail of Mt. Hallasan, male plants were dominant. Plants of A. aethusifolius showed a patchy distribution pattern in the population. A clump consisted of two female plants (Fig. 1A), one clump contained three male and four andropolygamous plants (Fig. 1E), and the remaining clumps included male plants. Our field observation also shows that the frequency of andro-polygamous plants in A. aethusifolius is higher than that in A. dioicus.
Why does A. aethusifolius show a high frequency of andropolygamous plants? It may be that the fitness of bisexual flowers, the population size, the genetic mechanism of sex determination, or a combination of these factors is associated with the frequent development of bisexual flowers. The occurrence of the same pattern of sexuality in all taxa of Aruncus, including A. dioicus and A. aethusifolius, suggests that polygamo-dioecious is genetically determined. Given that its sister group, Holodiscus, and most other members of Spiraeeae are hermaphroditic (Table 1), the gender polymorphism and underlying genetic network in A. aethusifolius should have evolved in the most recent common ancestor of Aruncus. It is interesting to note that seeds produced by bisexual flowers grow into andro-polygamous plants in A. dioicus var. dioicus (Bond, 1962). If we assume that this system applies to A. aethusifolius, the genotype of the andropolygamous plant is inherited and maintained in a population. The frequency of the genotype in A. aethusifolius may be high due to the large population size on Mt. Hallasan and indeed on Jeju Island. The large population size compared to A. dioicus may have played a role in the preservation of the genotype of the andro-polygamous plant.
The fitness of the andro-polygamous plants in A. aethusifolius may be greater than the fitness of andropolygamous plants in A. dioicus. Bisexual flowers are borne on the part of the male inflorescence, as described above (Fig. 2). Thus, the proportion of seeds from andro-polygamous plants in a population would decrease as the size of the inflorescence increases. The size of the panicles of female plants in A. dioicus is very large (11–27.8 cm in length [mean = 17.5 cm]), producing numerous follicles. In contrast, the size of the inflorescence of female plants in A. aethusifolius is small, ranging from 7.7 to 12.5 cm in length (mean = 9.4 cm). This suggests that the magnitude of the difference in the number of seeds produced between carpellate and bisexual flowers in A. aethusifolius is smaller compared to that in A. dioicus. This would result in a higher proportion of seeds from bisexual flowers in a seed pool or bank in a population of A. aethusifolius. Further study of the gender specificity of these seeds is therefore necessary.
The genetic mechanisms which determine gender in plants are diverse (Pannell, 2017) because dioecy evolved multiple times from hermaphroditism. A genetic network for sex determination controlled by environmental signals was shown in Ceratopteris richardii Brongn. (Tanurdzic and Banks, 2004). Chromosomal sex determination has been widely studied in several angiosperms, such as Fragaria virginiana Mill. (Spigler et al, 2008), Sagittaria latifolia Willd. (Dorken and Barret, 2004), and Silene latifolia Poir. (Fujita et al., 2012). In monoecious plants such as melons and maize, the sexual identity of flowers as either male or female is determined by different genetic networks that react to hormones (Irish, 1999; Boualem et al., 2015). These case studies suggest that there is no general model of gender determination in plants. It would be interesting and important to study the sex determination mechanism in A. aethusifolius.